A Randomized Trial of the Amikacin Fosfomycin Inhalation System for the Adjunctive Therapy of Gram‐Negative Ventilator‐Associated Pneumonia: IASIS Trial | Semantic Scholar

PDF) Amikacin nebulization for the adjunctive therapy of gram-negative pneumonia in mechanically ventilated patients: a systematic review and meta-analysis of randomized controlled trials

A comparative study between effect of combined intravenous and nebulized amikacin versus intravenous amikacin alone in mechanically ventilated patients with ventilator-associated pneumonia (VAP) | Ain-Shams Journal of Anesthesiology | Full Text

Pharmacokinetics of high-dose nebulized amikacin in mechanically ventilated healthy subjects | Intensive Care Medicine

The Unfulfilled Promise of Inhaled Therapy in Ventilator-Associated Infections: Where Do We Go from Here? | Journal of Aerosol Medicine and Pulmonary Drug Delivery

Mean cumulative systemic absorption of amikacin as a function of time.... | Download Scientific Diagram

Amikacin Liposome Inhalation Suspension for Refractory Mycobacterium avium Complex Lung Disease: Sustainability and Durability of Culture Conversion and Safety of Long-term Exposure - ScienceDirect

Delivery Efficacy of a Vibrating Mesh Nebulizer and a Jet Nebulizer under Different Configurations | Journal of Aerosol Medicine and Pulmonary Drug Delivery

Antibiotics | Free Full-Text | Pharmacokinetic Characteristics of Nebulized Colistimethate Sodium Using Two Different Types of Nebulizers in Critically Ill Patients with Ventilator-Associated Respiratory Infections

Inhaled amikacin adjunctive to intravenous standard-of-care antibiotics in mechanically ventilated patients with Gram-negative pneumonia (INHALE): a double-blind, randomised, placebo-controlled, phase 3, superiority trial - The Lancet Infectious Diseases
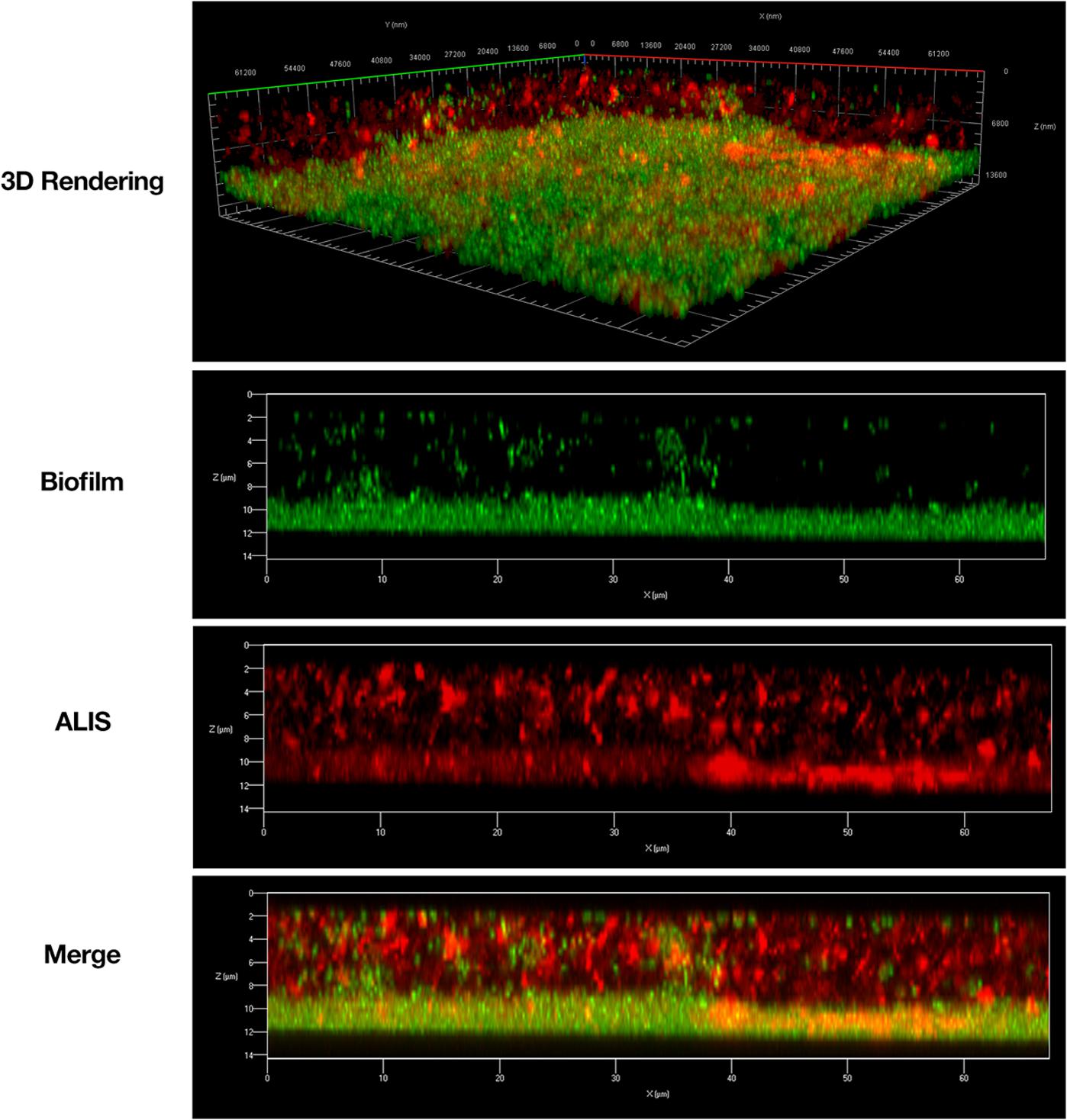
Frontiers | Amikacin Liposome Inhalation Suspension (ALIS) Penetrates Non-tuberculous Mycobacterial Biofilms and Enhances Amikacin Uptake Into Macrophages

Amikacin nebulization for the adjunctive therapy of gram-negative pneumonia in mechanically ventilated patients: a systematic review and meta-analysis of randomized controlled trials | Scientific Reports

Amikacin Inhalation as Salvage Therapy for Refractory Nontuberculous Mycobacterial Lung Disease | Antimicrobial Agents and Chemotherapy

Frontiers | Amikacin Liposome Inhalation Suspension (ALIS) Penetrates Non-tuberculous Mycobacterial Biofilms and Enhances Amikacin Uptake Into Macrophages
PARI Pharma's eFlow® Technology device, LAMIRA™, approved as the only nebulizer system to deliver Insmed's ARIKAYCE® (amikacin liposome inhalation suspension)